  
So the small molecule C 2H 4 is not an alkane. Here's the equation again: C 10H 22(g) C 8H 18(g) + C 2H 4(g) Remember that the general formula for an alkane is C nH (2n+2). Molecule formed in the equation given for cracking in the previous section? 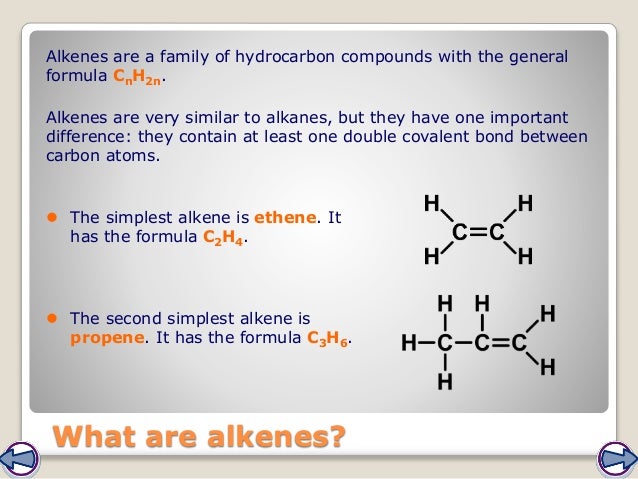
MoleculeA molecule is a group of two or more atoms bonded together. (A catalyst is a substance that speeds up a chemical reaction but remains chemically unchanged itself at the end of the reaction.) Look at an example of cracking below: C 10H 22(g) C 8H 18(g) + C 2H 4(g) Cracking is an example of a thermal decomposition reaction (in this case of an. Catalyst, and they break down into smaller molecules. CatalystA catalyst is a substance that alters (usually speeds up) the rate of a chemical reaction, but remains chemically unchanged itself at the end of the reaction. But we just don't use so much of the heavier fractions, so chemists have found ways to meet the demand for petrol by producing this from the heavier hydrocarbons. The fractions that provide us with fuel for cars are in great demand. 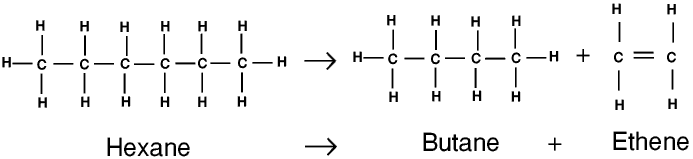
As you might expect, we do not need as much of some fractions as others. It is the raw material for many fuels and plastics. Crude oilCrude oil is a liquid formed from tiny sea creatures and plants that died millions of years ago. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
